March was full of novelties! WHO published a report on the access to new medicines in Europe and the EEA published the State of the Environment linking environmental policies to our health. The Commission announced a prize for alternative solutions to the use of antibiotics and the European Patients Forum (EPF) adopted its positions on adherence/concordance and on the communication of clinical trials results to patients. WHO report on the access to the new medicines in Europe The World Health Organisation (WHO) has published a report with a focus on sustainable access to new medicines, reviews of policies that affect medicines throughout their lifecycle (from research and development to disinvestment). Taking stock of the current evidence across Europe, the report highlights that the number of new medicines introduced in Europe is increasing, especially for chronic diseases such as cancers and COPD.
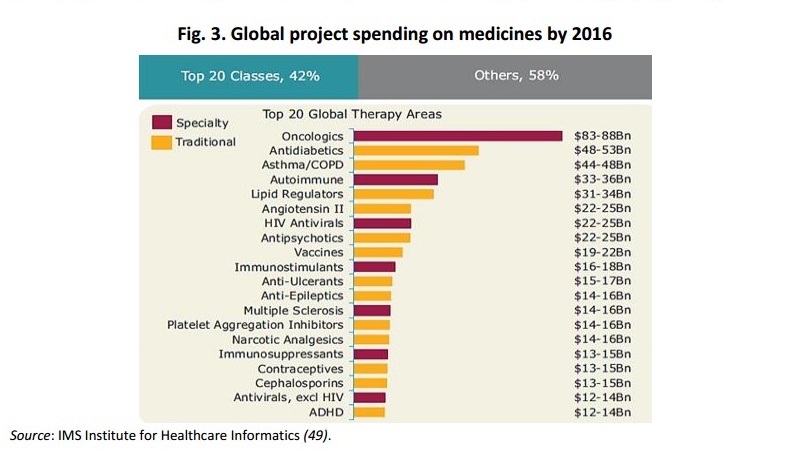 The report features findings from 27 countries and explores different ways that health authorities in European countries are dealing with high spending on new medicines, including methods such as restrictive treatment guidelines, target levels for use of generics, and limitations on the use of particularly expensive drugs. The report notes that pharmaceutical companies often aim to charge higher prices for newer drugs to recover investment costs in research and development. With this report, WHO outlines possible policy directions and choices that may help governments to reduce high prices when introducing new drugs.
The report features findings from 27 countries and explores different ways that health authorities in European countries are dealing with high spending on new medicines, including methods such as restrictive treatment guidelines, target levels for use of generics, and limitations on the use of particularly expensive drugs. The report notes that pharmaceutical companies often aim to charge higher prices for newer drugs to recover investment costs in research and development. With this report, WHO outlines possible policy directions and choices that may help governments to reduce high prices when introducing new drugs.
To read the full report please visit WHO website. Antimicrobial resistance – a 1 million Euro prize for the alternative cure The European Commission has recently launched the Horizon Prize for Better Use of Antibiotics. It is a €1 million challenge prize that will be granted to the best test: cheap, rapid, easy-to-use for healthcare providers and non- or minimally invasive for patients with upper respiratory tract infections that can be treated safely without antibiotics. The objective is to stop overuse of antibiotics and halt the growing antimicrobial resistance. According to data from 2011, 25,000 patients die annually as a result of infections caused by resistant bacteria in the EU. The costs incurred by drug resistant infections amount to an estimated €1.5 billion annually, due to increases in healthcare expenditure costs and productivity losses. Those who are ready to come up with the most promising and effective solution are welcome to apply for the European Commission’s prize from 10 March 2015 to 17 August 2016 and the winner of the prize will be known at the end of 2016. If you wish to participate, please visit the Horizon Prize page. EPF Position on Adherence and Concordance In March, the European Patients’ Forum (EPF) issued a position paper on adherence and concordance that includes EFA’s views. The aim of this document is to clarify the terminology of adherence from the patients’ perspective. The topic is firmly positioned on patient-centred healthcare, patient empowerment, shared decision-making, information and health literacy. Effective use of medicines is an important part of self-management of chronic conditions. However, many patients do not take their medicines as intended by the prescriber. This can be for a number of reasons, not all of them to do with the individual patient or the treatment itself. Non-adherence is an important patient safety issue that carries enormous costs, to patients personally in terms of avoidable ill health or extra treatment, and also to the health systems. Strategies to promote adherence should encompass health literacy, user-friendly information, joined-up healthcare teamwork, and self-management support. Health professionals should be trained in shared decision-making and in communicating effectively with patients about treatment choices and their personal, as well as medical, goals. If you wish to know more about adherence and concordance please read EPF’s position. EPF position on Clinical trial results – communicating the results to patients Also in March, the European Patients Forum (EPF) published its position on clinical trials results. The new EU Clinical Trials Regulation (Regulation 536/2014) introduces a number of improvements to the transparency of clinical trials results:
- Compulsory registration of all trials;
- Principle of public access to the EU clinical trials database developed and maintained by the European Medicines Agency;
- Publication of all trial results, irrespective of the outcomes, on the EU database;
- Summary results must include a summary “understandable to lay person”.
Thanks to the new regulation, the summary results including lay summary, will be published within one year after the end of the trial, irrespective of the outcomes of the trial. What is more, patients participating in the clinical trial must be informed on the availability of the lay summary of the results. In the position paper, EPF calls for a set of guidelines to be developed at EU level, with the involvement of patient organisations, to ensure that the lay summaries written for the EU database adhere to a common set of quality standards. In this sense, the existing guidance on lay summaries should be reviewed and used to arrive at the common set of guidelines. It is suggested that the guidelines should be drafted by a dedicated working group hosted at the European Medicines Agency, including patient representatives, the European Commission and industry. The full EPF position can be accessed here. Environment report shows benefits of EU action 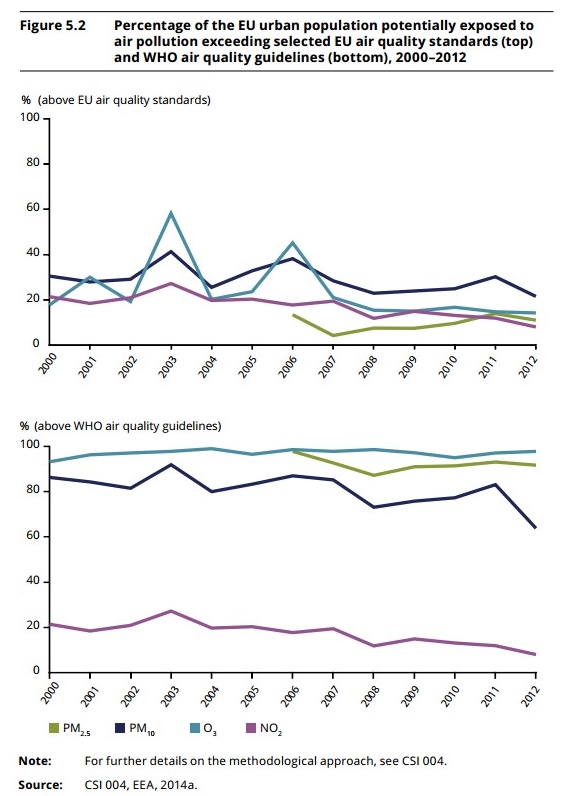 A new report prepared by the European Environment Agency (EEA) called State and Outlook 2015. The report contains an integrated assessment of Europe’s environment that also includes assessments and data at global, regional and country levels, as well as cross-country comparisons. Health and environment has a dedicated chapter with risks for human health posed by environmental determinants. The report shows that a coordinated EU environment policy has delivered substantial benefits for citizens over the past five years. The report states that Europeans enjoy cleaner air and water, send less waste to landfill and recycle more; while environmental policies drive jobs and growth. But the report warns that Europe's long-term goal of "living well, within the limits of our planet" – the aim of the Seventh Environment Action Programme (7th EAP) – cannot be achieved with the current level of ambition of environment and related policies. The analysis calls for more integrated policy making – a call the Commission is responding to with a series of environmental initiatives for 2015, including a new broader package on the Circular Economy, a review of the EU Biodiversity Strategy, an action plan on Ocean Governance and a modified Air Quality package. EFA is advocating at EU level for levels of air quality, both indoors and outdoors, that do not harm human health and for reducing chemicals' exposure in everyday life. If you are interested to know more about environmental policies and their impact on our health, please read the 2015 SOER report. EMPATHIE project results For about a year, the tender ‘Empowering patients in the management of chronic diseases’ (or EMPATHiE) analysed empowerment for patients with chronic diseases. Among the key findings, EMPATHiE confirmed that interventions targeting patient empowerment tend to present positive results (when compared to usual care). Another good news is the strong interest patient empowerment generates: all stakeholders involved in the study demonstrated their willingness to collaborate at European level on patient empowerment.
A new report prepared by the European Environment Agency (EEA) called State and Outlook 2015. The report contains an integrated assessment of Europe’s environment that also includes assessments and data at global, regional and country levels, as well as cross-country comparisons. Health and environment has a dedicated chapter with risks for human health posed by environmental determinants. The report shows that a coordinated EU environment policy has delivered substantial benefits for citizens over the past five years. The report states that Europeans enjoy cleaner air and water, send less waste to landfill and recycle more; while environmental policies drive jobs and growth. But the report warns that Europe's long-term goal of "living well, within the limits of our planet" – the aim of the Seventh Environment Action Programme (7th EAP) – cannot be achieved with the current level of ambition of environment and related policies. The analysis calls for more integrated policy making – a call the Commission is responding to with a series of environmental initiatives for 2015, including a new broader package on the Circular Economy, a review of the EU Biodiversity Strategy, an action plan on Ocean Governance and a modified Air Quality package. EFA is advocating at EU level for levels of air quality, both indoors and outdoors, that do not harm human health and for reducing chemicals' exposure in everyday life. If you are interested to know more about environmental policies and their impact on our health, please read the 2015 SOER report. EMPATHIE project results For about a year, the tender ‘Empowering patients in the management of chronic diseases’ (or EMPATHiE) analysed empowerment for patients with chronic diseases. Among the key findings, EMPATHiE confirmed that interventions targeting patient empowerment tend to present positive results (when compared to usual care). Another good news is the strong interest patient empowerment generates: all stakeholders involved in the study demonstrated their willingness to collaborate at European level on patient empowerment.
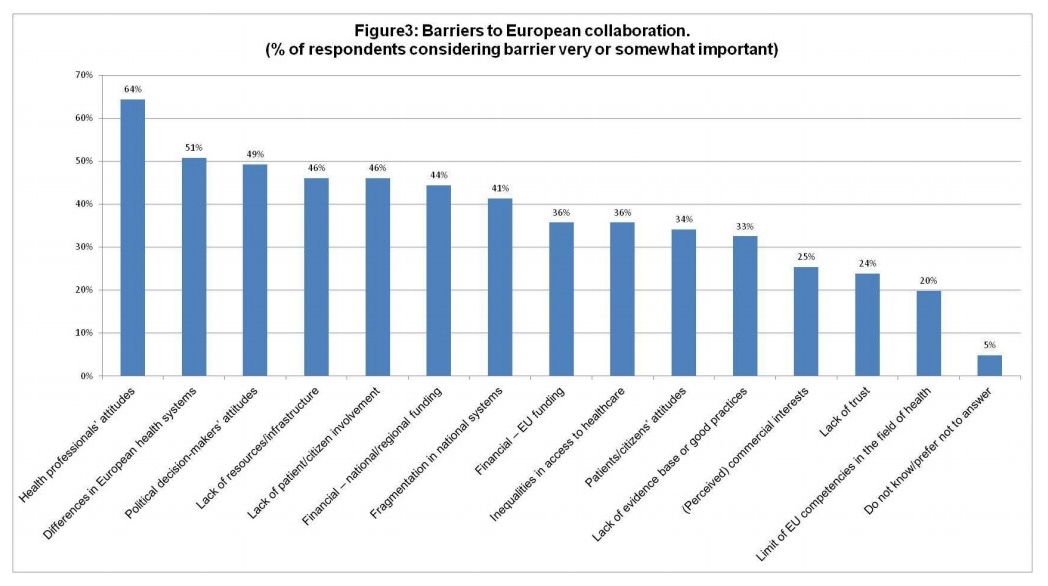 The study recommendations are to develop a European strategy and action plan on patient empowerment as a starting point and to:
The study recommendations are to develop a European strategy and action plan on patient empowerment as a starting point and to:
- Provide better education to patients and the wider audience;
- Improve the education of healthcare professionals in holistic thinking and communication;
- Restructure healthcare delivery and develop a central common electronic record accessible by patients as well as professionals.
To know more about EMPATHiE results please visit the study website.